
A scientific study partially funded by the Life Extension Foundation has demonstrated that the aging of human cells can be reversed. Published in the journal Regenerative Medicine, researchers succeeded for the first time in turning back the clock of aging in adult human cells. In an exclusive interview, Dr. Michael West explains the results of this unprecedented breakthrough.
Scientifically reviewed by: Dr. Gary Gonzalez, MD, in January 2021. Written by: Gregory M. Fahy, PhD and Saul Kent.
n Feb. 20, 2010, Gregory M. Fahy, PhD and Saul Kent interviewed Michael West, PhD, CEO of BioTime, Inc., about a new breakthrough published in the journal Regenerative Medicine. The paper reported the reversal of what Dr. West has called the “developmental aging” of adult human cells in the laboratory dish. Utilizing genes that grant our reproductive cells the potential for immortal growth, the researchers showed that it was possible to turn back the clock in human body cells, enabling the potential for young patient-specific cells of any kind for use in regenerative medicine. This research was funded in part by the Life Extension Foundation®. We asked Dr. West to elaborate on the details of his groundbreaking research, and to expound on the implications for the future of rejuvenative medicine.Fahy: Before we get to the details of your paper on the reversal of developmental aging, let’s set the stage. Your discovery relates to the field of regenerative medicine. What do you mean by regenerative medicine and how does it differ from medicine as it exists today?West: Well, the name “regenerative medicine” came from Bill Haseltine, then of Human Genome Sciences, one of the early leaders in genomics and DNA technology. Back in the 1990s, Bill learned that researchers in aging were making important progress on turning back the clock of aging in human cells through cloning, and then creating young cells that could potentially regenerate or repair all the tissues of the aged human body. And so, upon hearing of that realistic prospect, he christened the field “regenerative medicine” in the belief that it would one day become a major part of medical practice. So, based on its origins, I would define regenerative medicine as that collection of technologies that utilizes embryonic pluripotent stem cells and their derivatives to regenerate tissues in the body ravaged from disease, primarily degenerative disorders associated with aging.Click to enlargeFahy: The implication of the term is that we’re actually going to be able to regenerate or re-grow parts of the aging body because of this ability to turn back the clock of cellular aging.West: Yes. First, let’s talk about cellular aging. The problem with human biology is that the immortal reproductive cells that built you and me develop into differentiated cells within our bodies and as a result, lose the capacity to proliferate (divide) forever. So, the cells of the body are mortal, meaning they have a finite life span, and as our tissues age, or deteriorate from disease, our body has a finite capacity to regenerate and repair those tissues. As a result, we suffer progressive declines in function that lead to our death.Fahy: So, are you saying that an inadequate ability to generate new cells in the aging body is linked to the body’s inability to repair itself as we grow older?West: Every tissue is different, but that’s basically what I am saying. The goal of gerontology for many years has been to find the reason that our reproductive lineage continues to make babies generation after generation while the other cells in our body, called somatic cells, have a finite life span and are mortal, or, in other words, to discover the reason babies are born young. The answer is that we come from a lineage of cells that have been proliferating since the dawn of life on earth. The cells that made us have no dead ancestors. That recognition is causing a major shift in our thinking about aging, towards recognizing that aging may be more simple than we once thought. Although complex in the way it’s played out in thousands of genes and proteins, in reality, it may be that only a small number of central mechanisms cause the somatic cells in our body to age. So, the goal has been to discover a way of transferring the immortality of reproductive cells into the body in order to increase the potential life span of individual human beings.Fahy: What gives the germ line immortality, or as you put it, why are babies born young?West: Well, the first clue was published in 1986.1 Howard Cooke reported that specific regions of DNA at the ends of our chromosomes called telomeres are long and constant in length in our reproductive cells (sperm in particular in this case) with age, but shorten progressively in body cell types as we age.That publication led me to realize that an old theory of aging by Alexey Olovnikov from Russia now had scientific support. Olovnikov’s theory was that the difference between our reproductive cells and our body cells is that the ends of the DNA strands shorten over time in somatic or body cells, but are maintained at a long length in reproductive cells.2That shortening was considered by Olovnikov to be a clock of cellular aging, similar to the way the length of a fuse leading to a bomb can be a kind of clock—the longer it’s set, the longer it will burn before the bomb goes off. And, of course, the bomb going off here is the aging of somatic cells. Olovnikov proposed that there is an immortalizing enzyme that is shut off in body cell types, causing them to have a finite life span.Back around 1990, I became convinced the telomere hypothesis was correct, and founded Geron, where we isolated and purified this immortalizing enzyme, which we called telomerase. In 1998 we demonstrated that the addition of telomerase to body cell types such as skin cells or cells from the retina involved in macular degeneration stopped these cells from aging.3IMMORTAL CELLSEarly in the history of evolution, life existed as single cells, not unlike the protozoa swimming around in pond water today. These animals replicated by simply splitting into two new cells. They didn’t have to die and are therefore called “immortal.”In the following millennia, these immortal cells spun off specialized helper cells to help them compete in feeding and reproduction. These helper cells selflessly served the needs of the immortal cells and became what we call the “body” while the immortal cells became what we call the “germ line.” Since the immortal cells carried genetic information, they selected for the body to die after it served its purpose.Where are these immortal cells in you and me? In the adult human, they are the egg cells in a woman’s ovary, and the sperm-forming cells in the testicles of a man. When a sperm and egg unite, the resulting cells continue the germ line by forming a small cluster of immortal cells that go on to make new body and new immortal reproductive cells of a new human being, a cycle that continues forever.For the first time in the history of life on earth, the body cells have evolved a brain that is capable of understanding evolution and capable of deciphering the molecular mechanisms of cellular mortality and immortality. This conscious body is now plotting to take on the legacy of immortality for itself.Fahy: What are adult stem cells?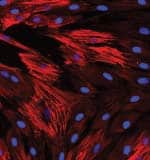 Embryonic stem cells can turn into all the cells of the human body. Here, smooth muscle cells are stained red while the nuclei are stained blue.West: There are tissues in the human body that have evolved a source of regenerative cells—or adult stem cells—to repair the tissues when they are damaged. For example, when a cave bear attacked an ancient Neanderthal and scratched the surface of his skin, there had to be stem cells there that could within a few days repair that damage. Similarly, our livers can regenerate extensively when damaged by toxins, accidents, or diseases. And when you have a strained muscle, muscle can regenerate too.But unfortunately, not all cells and tissues of the human body have extensive regenerative capacity, and indeed, in the case of humans, adult stem cells all appear to be mortal. They can regenerate tissue function, but only for a finite period of time. We can observe the aging of adult stem cells through telomere shortening.If we can learn the lessons of how our reproductive lineage has been creating babies for millions of years to continue the human species, we should be able to design medical therapies to allow the human body to regenerate itself and escape the genetic boundaries of human life.Fahy: You have suggested in the past that this might be accomplished using stem cells derived from embryos, and, in particular, embryos created by somatic cell nuclear transfer (cloning).West: Once we knew it was telomerase that was maintaining the germ line, it occurred to me that we might be able to isolate germ line cells from human embryos and just propagate them in a laboratory dish. These cells should be immortal in the laboratory just like they are in nature. Think of it this way: they would be in a sense the immortal stem cell of human life itself, regenerating human beings generation after generation, forever. If we could tap into that amazing lineage of cells, it could allow us to manufacture all cell types in the human body. And theoretically we could make an unlimited assembly line, making new young tooth-forming cells, new young cells for the retina, for the cornea, for the heart, for the kidneys, and so on. And so that was the genesis of the project to isolate human embryonic stem cells, a project that began in the mid-1990s and culminated with the publication of human embryonic stem and embryonic germ cells in 1998.4,5The isolation of these cells was controversial because of the need to derive the cells from human embryos. Certainly elevating their notoriety was President George W. Bush’s first national address on August 9, 2001, when he discussed the ethics of the cells and policy on federal funding of the research.These cells nevertheless caused a great deal of enthusiasm. I think people were initially assuming that existing embryonic stem cell lines would be used for the manufacture of all future products. But in my mind, those lines couldn’t ultimately work for many uses because of transplant rejection. Many of the cell types in the human body are rejected after being transplanted from one person to another. The immune system recognizes them as foreign cells and attacks and kills them. So we initially sought to solve the problem of transplant rejection by making these all-powerful embryonic stem cells genetically identical to the patient in need of the therapy through cloning.In 2001, we published the first account of cloned human embryos in an attempt to make patient-specific stem cells.6 We said at the time that our goal was not the cloning of human beings, but was only to clone patient-specific embryonic stem cells that would not be rejected. We anticipated that the use of cloning would not only take a skin cell back to an embryonic state, but that it would also make an old cell young again by rebuilding the telomere back to an embryonic state. So our goal was to be able to produce young cells of any type identical to the patient’s own cells, and we called the process for doing this therapeutic cloning.7Fahy: Why was therapeutic cloning insufficient?West: Of course, it was very controversial because of the need to clone human embryos. In addition, the difficulties of getting a sufficient number of human egg cells and the technical challenge of implementing cloning made advances in that field slow and difficult. What we desperately needed was to find a means to make all these therapies we’d been talking about possible on a scalable and affordable platform that would allow them to be implemented commercially for millions of patients.Fahy: Which brings us to induced pluripotent stem cells (or iPS cells). What are these cells and how are they generated?West: iPS cell technology is a way of using just a handful of genes to coax a cell back in time without using egg cells or cloning. And this is the exciting part: the technology works. It is one of the most active areas in medical research today.Let me tell you a fun story. Shortly after we cloned human embryos in 2001, I was at a birthday party for Jack Szostak in Boston. Jack, along with Liz Blackburn and Carol Greider, won the Nobel Prize in 2009 for their early research in telomere biology. Mark Ptashne was at Jack’s house too, and after a few glasses of wine I asked him, “Mark, after hearing about cloning, how many genes do you think are in the egg cell that are responsible for taking the cell back in time?” After a couple more sips of wine, Mark, who is an expert in transcription factors, the master regulators of all the genes in somatic cells, said quite definitely, though I think tongue-in-cheek, “four.” I responded “you’re wrong” with an exclamation point, because I believed there were published data suggesting that what is in the egg cell that is required to turn back the clock in cells is enormously complex.However, a couple years later we were using microarray DNA sequences on a chip to look at the thousands of genes in embryonic stem cells and in hundreds of cell types we had purified using a technology we call ACTCellerate. And using computer programs to search for the genes that were different in these cells, we found, at the top of the list, almost as if highlighted in glowing bold letters, four transcription factors that are active in embryonic stem cells and inactive in virtually all body cell types—Oct4, Sox2, Lin28, and Nanog. I looked at these genes and, remembering my conversation with Mark, had to ask myself, “Could it really be that simple? Could I be looking at the master regulators of the immortal renewal of human life?”As I thought about this carefully, I came to believe that the earlier data that cloning was complex was not that solid, and so we filed patent applications on the use of these and other related germ line-specific genes as master regulators that, if expressed in a human body cell, would be fully capable of transporting an old skin cell (and many other adult body cells) back to an embryonic state, rewinding the “clock” of development whereby embryonic cells normally differentiate into the specialized cells of the body, as well as the telomere “clock” of cellular aging. Since this is resetting both, we call it the reversal of developmental aging.Kent: You’re leaving out the fact that scientists did, in fact, find that these four transcription factors do indeed work. Embryonic stem cells can turn into all the cells of the human body. Here, smooth muscle cells are stained red while the nuclei are stained blue.West: There are tissues in the human body that have evolved a source of regenerative cells—or adult stem cells—to repair the tissues when they are damaged. For example, when a cave bear attacked an ancient Neanderthal and scratched the surface of his skin, there had to be stem cells there that could within a few days repair that damage. Similarly, our livers can regenerate extensively when damaged by toxins, accidents, or diseases. And when you have a strained muscle, muscle can regenerate too.But unfortunately, not all cells and tissues of the human body have extensive regenerative capacity, and indeed, in the case of humans, adult stem cells all appear to be mortal. They can regenerate tissue function, but only for a finite period of time. We can observe the aging of adult stem cells through telomere shortening.If we can learn the lessons of how our reproductive lineage has been creating babies for millions of years to continue the human species, we should be able to design medical therapies to allow the human body to regenerate itself and escape the genetic boundaries of human life.Fahy: You have suggested in the past that this might be accomplished using stem cells derived from embryos, and, in particular, embryos created by somatic cell nuclear transfer (cloning).West: Once we knew it was telomerase that was maintaining the germ line, it occurred to me that we might be able to isolate germ line cells from human embryos and just propagate them in a laboratory dish. These cells should be immortal in the laboratory just like they are in nature. Think of it this way: they would be in a sense the immortal stem cell of human life itself, regenerating human beings generation after generation, forever. If we could tap into that amazing lineage of cells, it could allow us to manufacture all cell types in the human body. And theoretically we could make an unlimited assembly line, making new young tooth-forming cells, new young cells for the retina, for the cornea, for the heart, for the kidneys, and so on. And so that was the genesis of the project to isolate human embryonic stem cells, a project that began in the mid-1990s and culminated with the publication of human embryonic stem and embryonic germ cells in 1998.4,5The isolation of these cells was controversial because of the need to derive the cells from human embryos. Certainly elevating their notoriety was President George W. Bush’s first national address on August 9, 2001, when he discussed the ethics of the cells and policy on federal funding of the research.These cells nevertheless caused a great deal of enthusiasm. I think people were initially assuming that existing embryonic stem cell lines would be used for the manufacture of all future products. But in my mind, those lines couldn’t ultimately work for many uses because of transplant rejection. Many of the cell types in the human body are rejected after being transplanted from one person to another. The immune system recognizes them as foreign cells and attacks and kills them. So we initially sought to solve the problem of transplant rejection by making these all-powerful embryonic stem cells genetically identical to the patient in need of the therapy through cloning.In 2001, we published the first account of cloned human embryos in an attempt to make patient-specific stem cells.6 We said at the time that our goal was not the cloning of human beings, but was only to clone patient-specific embryonic stem cells that would not be rejected. We anticipated that the use of cloning would not only take a skin cell back to an embryonic state, but that it would also make an old cell young again by rebuilding the telomere back to an embryonic state. So our goal was to be able to produce young cells of any type identical to the patient’s own cells, and we called the process for doing this therapeutic cloning.7Fahy: Why was therapeutic cloning insufficient?West: Of course, it was very controversial because of the need to clone human embryos. In addition, the difficulties of getting a sufficient number of human egg cells and the technical challenge of implementing cloning made advances in that field slow and difficult. What we desperately needed was to find a means to make all these therapies we’d been talking about possible on a scalable and affordable platform that would allow them to be implemented commercially for millions of patients.Fahy: Which brings us to induced pluripotent stem cells (or iPS cells). What are these cells and how are they generated?West: iPS cell technology is a way of using just a handful of genes to coax a cell back in time without using egg cells or cloning. And this is the exciting part: the technology works. It is one of the most active areas in medical research today.Let me tell you a fun story. Shortly after we cloned human embryos in 2001, I was at a birthday party for Jack Szostak in Boston. Jack, along with Liz Blackburn and Carol Greider, won the Nobel Prize in 2009 for their early research in telomere biology. Mark Ptashne was at Jack’s house too, and after a few glasses of wine I asked him, “Mark, after hearing about cloning, how many genes do you think are in the egg cell that are responsible for taking the cell back in time?” After a couple more sips of wine, Mark, who is an expert in transcription factors, the master regulators of all the genes in somatic cells, said quite definitely, though I think tongue-in-cheek, “four.” I responded “you’re wrong” with an exclamation point, because I believed there were published data suggesting that what is in the egg cell that is required to turn back the clock in cells is enormously complex.However, a couple years later we were using microarray DNA sequences on a chip to look at the thousands of genes in embryonic stem cells and in hundreds of cell types we had purified using a technology we call ACTCellerate. And using computer programs to search for the genes that were different in these cells, we found, at the top of the list, almost as if highlighted in glowing bold letters, four transcription factors that are active in embryonic stem cells and inactive in virtually all body cell types—Oct4, Sox2, Lin28, and Nanog. I looked at these genes and, remembering my conversation with Mark, had to ask myself, “Could it really be that simple? Could I be looking at the master regulators of the immortal renewal of human life?”As I thought about this carefully, I came to believe that the earlier data that cloning was complex was not that solid, and so we filed patent applications on the use of these and other related germ line-specific genes as master regulators that, if expressed in a human body cell, would be fully capable of transporting an old skin cell (and many other adult body cells) back to an embryonic state, rewinding the “clock” of development whereby embryonic cells normally differentiate into the specialized cells of the body, as well as the telomere “clock” of cellular aging. Since this is resetting both, we call it the reversal of developmental aging.Kent: You’re leaving out the fact that scientists did, in fact, find that these four transcription factors do indeed work.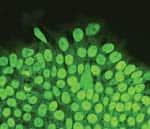 The age-reversed cells in the study are shown here stained for a marker called “Oct4”, a protein normally only expressed in the immortal reproductive lineage.West: Right. In 2007, Yamanaka of Japan and James Thomson in Wisconsin, published the first papers showing that this family of transcription factors was indeed successful at turning back the developmental clock of aging.8,9 Taking a skin cell, for instance, back to an embryonic state. Since no embryos are used in their derivation, they are called “induced pluripotent stem (iPS) cells” instead of embryonic stem (ES) cells.Fahy: iPS cell technology has been on the cutting edge of regenerative medicine ever since. But you found a problem. What is it?West: Recognizing the problems of implementing cloning, we began working in this field in the 1999–2005 time frame with a strong interest in what happens to the clock of cellular aging. Thomson and Yamanaka had not carefully looked at whether the clock of cellular aging was reversed in their iPS cell lines, so after these lines and others were widely distributed in the scientific community, we surveyed their telomere lengths and saw that they were not at the length expected for the beginning of life.In cloning, when you take a cell from an old animal and transplant that cell or its nucleus into an egg cell, the clock of aging gets rewound. So even though you take a cell from a very old animal, the young cloned animal is born young. Cloning takes a body cell and transforms it back into an immortal germ line cell and creates a baby just as if it had been sperm and egg that created it. The rumors that cloned animals are generally born old are myths.10 That’s been firmly shown not to be the case.11 So the egg cell can function as a cellular time machine, reversing developmental aging,12 so an animal cloned from an old animal is born young.13Now, with iPS technology, which is a way of performing the equivalent of cloning without using an egg cell or making an embryo, everyone believed from the start that it would also transport the cell back to immortality, since that is what happens in cloning. But when we looked at telomeres in several widely used iPS cell lines we saw they were all short. In the paper we just published,14 our survey showed that, although all iPS cell lines out there do reactivate telomerase and otherwise look like embryonic stem cells, they do not properly reset the clock of aging because their telomeres remain shorter than they should be.The good news is that we found a way to identify iPS cells that have reset the clock of aging. And so while iPS cell technology is not as efficient as cloning in rapidly and reliably reversing the clock of aging, we’ve shown in this paper that it can be made to work quite simply. So the ability to reverse the aging of human cells both from the standpoint of embryological development and in terms of the clock of aging and in an ethically non-problematic manner, and to do it on a commercial and affordable scale, makes regenerative medicine an attractive pathway to profoundly intervene in human aging.Kent: But as you said, the majority of iPS cells don’t maintain the length of their telomeres. What percentage of the cells that are produced in this manner are, in fact, turned back in time based on their telomere lengths and telomerase activity?West: These are still the early days of our investigation of iPS technology, but of the six lines we generated, one successfully reset the clock of aging. And so our current success rate is one out of six. But of course those numbers will likely change with larger studies.Kent: Do you know any reason that might explain why one in six are different from the others?West: We’ve taken the successful cell line whose telomeres got reset all the way back to embryonic telomere lengths again, and looked at it carefully using DNA chips that measure gene expression in all human genes, and we were able to identify some genes related to the telomere that, at minimum, are a marker of the cells whose clocks have been turned back successfully.Why the egg cell is so much more efficient than these genes remains unknown, but the important thing is that it really is a matter of efficiency here. Even if the efficiency is never improved beyond one out of six, we still have a commercially feasible technology.Fahy: However, you referred in your paper to the possibility of developments such as the use of germ cell extracts and the like that might be able to improve the efficiency of making fully successful iPS cells. Do you have plans to actually pursue that, and is it a practical approach?West: Yes. We are now developing a technology we call ReCyte™. ReCyte™ uses testicular cells called EC cells that we genetically engineer to express large quantities of these master regulators such as Oct4, Sox2, and Lin28 to be little time machines to transport a patient’s cells back in time. ReCyte™ is a technology designed specifically to increase the efficiency of reprogramming, and to do so on a robotic, commercial scale. Based on our understanding of how all this technology works, we believe this will allow us to manufacture a product for resetting the clock of aging in cells inexpensively and efficiently and improve the quality of reprogramming, making the safest and most efficacious cells available for patients that can be produced as of today. The age-reversed cells in the study are shown here stained for a marker called “Oct4”, a protein normally only expressed in the immortal reproductive lineage.West: Right. In 2007, Yamanaka of Japan and James Thomson in Wisconsin, published the first papers showing that this family of transcription factors was indeed successful at turning back the developmental clock of aging.8,9 Taking a skin cell, for instance, back to an embryonic state. Since no embryos are used in their derivation, they are called “induced pluripotent stem (iPS) cells” instead of embryonic stem (ES) cells.Fahy: iPS cell technology has been on the cutting edge of regenerative medicine ever since. But you found a problem. What is it?West: Recognizing the problems of implementing cloning, we began working in this field in the 1999–2005 time frame with a strong interest in what happens to the clock of cellular aging. Thomson and Yamanaka had not carefully looked at whether the clock of cellular aging was reversed in their iPS cell lines, so after these lines and others were widely distributed in the scientific community, we surveyed their telomere lengths and saw that they were not at the length expected for the beginning of life.In cloning, when you take a cell from an old animal and transplant that cell or its nucleus into an egg cell, the clock of aging gets rewound. So even though you take a cell from a very old animal, the young cloned animal is born young. Cloning takes a body cell and transforms it back into an immortal germ line cell and creates a baby just as if it had been sperm and egg that created it. The rumors that cloned animals are generally born old are myths.10 That’s been firmly shown not to be the case.11 So the egg cell can function as a cellular time machine, reversing developmental aging,12 so an animal cloned from an old animal is born young.13Now, with iPS technology, which is a way of performing the equivalent of cloning without using an egg cell or making an embryo, everyone believed from the start that it would also transport the cell back to immortality, since that is what happens in cloning. But when we looked at telomeres in several widely used iPS cell lines we saw they were all short. In the paper we just published,14 our survey showed that, although all iPS cell lines out there do reactivate telomerase and otherwise look like embryonic stem cells, they do not properly reset the clock of aging because their telomeres remain shorter than they should be.The good news is that we found a way to identify iPS cells that have reset the clock of aging. And so while iPS cell technology is not as efficient as cloning in rapidly and reliably reversing the clock of aging, we’ve shown in this paper that it can be made to work quite simply. So the ability to reverse the aging of human cells both from the standpoint of embryological development and in terms of the clock of aging and in an ethically non-problematic manner, and to do it on a commercial and affordable scale, makes regenerative medicine an attractive pathway to profoundly intervene in human aging.Kent: But as you said, the majority of iPS cells don’t maintain the length of their telomeres. What percentage of the cells that are produced in this manner are, in fact, turned back in time based on their telomere lengths and telomerase activity?West: These are still the early days of our investigation of iPS technology, but of the six lines we generated, one successfully reset the clock of aging. And so our current success rate is one out of six. But of course those numbers will likely change with larger studies.Kent: Do you know any reason that might explain why one in six are different from the others?West: We’ve taken the successful cell line whose telomeres got reset all the way back to embryonic telomere lengths again, and looked at it carefully using DNA chips that measure gene expression in all human genes, and we were able to identify some genes related to the telomere that, at minimum, are a marker of the cells whose clocks have been turned back successfully.Why the egg cell is so much more efficient than these genes remains unknown, but the important thing is that it really is a matter of efficiency here. Even if the efficiency is never improved beyond one out of six, we still have a commercially feasible technology.Fahy: However, you referred in your paper to the possibility of developments such as the use of germ cell extracts and the like that might be able to improve the efficiency of making fully successful iPS cells. Do you have plans to actually pursue that, and is it a practical approach?West: Yes. We are now developing a technology we call ReCyte™. ReCyte™ uses testicular cells called EC cells that we genetically engineer to express large quantities of these master regulators such as Oct4, Sox2, and Lin28 to be little time machines to transport a patient’s cells back in time. ReCyte™ is a technology designed specifically to increase the efficiency of reprogramming, and to do so on a robotic, commercial scale. Based on our understanding of how all this technology works, we believe this will allow us to manufacture a product for resetting the clock of aging in cells inexpensively and efficiently and improve the quality of reprogramming, making the safest and most efficacious cells available for patients that can be produced as of today.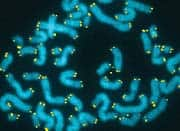 The clock of cellular aging resides at the very ends of the chromosomes, regions called “telomeres.” Shown here in a blue color are stained human chromosomes, and the telomeric DNA is stained bright yellow.Kent: You may be right about that, but right now the best you can do is one out of six. And as I understand it, the way this technology will be most effectively used is to take somatic cells from individuals and turn them back into embryonic-like cells. What is the chance that you’ll be unable to do so effectively in a particular individual?West: In our paper we manually created these lines, so we only made six cell lines, one of which properly rewound the clock. With the newer robotic platform we are developing, we could increase that number of lines markedly. So, even with no improvements in efficiency, we could increase the number of lines to make a successful outcome for a particular patient highly probable.Fahy: Is the ReCyte™ process patented by BioTime?West: Yes. ReCyte™ is a proprietary technology of BioTime and is based on several issued and pending patents.Fahy: Do you also have exclusive ownership over the process of choosing cells that spontaneously maintain their telomeres and excluding those that don’t?West: We’ve filed for patent protection on the selection process to identify these cell types based on the data in our paper.Fahy: Are there alternative ways of achieving the same effect you’ve been able to obtain? For example, it has been possible to artificially restore telomerase to cells since 1998,3 as you referred to earlier in the interview. Is there any reason iPS cells could not simply be telomerized to correct their defect in telomere maintenance?West: Well, we looked at a telomerized line in our publication, an iPS cell line from Harvard Medical School. That line had, in addition to these germ line genes that make this time machine work embryologically, some added telomerase, in the belief that there may be a problem with the telomere. But those cells still have inappropriately short telomeres. And so what we’ve learned is that telomerase is indeed very important, but merely adding telomerase to the iPS techniques as currently used in most laboratories is not sufficient.Fahy: In the paper, you speculate that maybe the reason the iPS cells are not maintaining their telomeres is that they’re not maintaining their undifferentiated embryonic state over time. Is there any evidence that they might be losing their undifferentiated state?West: All I can say is that what’s so striking about iPS cells is how similar they are to human embryonic stem cells in every respect. If one scans through 30,000 plus genes and looks at the expression of all those genes in human embryonic stem cells and iPS cells they are essentially indistinguishable. It’s very difficult, in fact, to find any differences, and it was hard for us to find what differences might be causing this inefficient resetting of the clock of aging. So the striking thing is their similarity, not that there are problems with the technology. However, we mention in the paper that our survey of widely-studied iPS cell lines may have been influenced by the poor culture of the cells in some laboratories.There have been reports that iPS cells are less likely to make various cell types, like neurons, compared to embryonic stem cells. But it’s important to remember that most of the world is working with iPS cells that are geriatric in terms of aging, and so all of those studies need to be repeated with these new cell lines that have their clock of aging reset properly before any conclusions can be drawn about the normality or abnormality of iPS cells.Fahy: What about the possibility that the cells that are naturally able to restore and maintain their telomeres are those that have DNA damage?West: We looked carefully throughout the genome of these cells using what is called SNP analysis and couldn’t find any evidence of DNA damage such as rearrangements, so for this and other reasons, we don’t have any reason to think the iPS cells that maintain their telomeres do so because of DNA damage the way cancer cells do.A long-standing theory of aging is that our body accumulates cells that have broken chromosomes, serious DNA damage, and damage unrelated to telomere shortening per se. And that almost certainly happens. Nevertheless, we also know that the human body has many trillions of cells that are completely intact, and with modern sequencing technologies it’s now possible for just a few thousand dollars to completely sequence a cell line’s genome. And so I believe it will be commercially feasible to perform a previously unthinkable amount of quality control to be sure that the young cell types that we make by new technologies like ReCyte™ are pristine, young cells as good as the cells we were born from, and not carrying a significant load of genetic damage.Kent: Robert Bradbury and others have talked about plucking pristine stem cells out of a pool of adult stem cells and having the best possible adult stem cells rather than some of them being contaminated or damaged.West: There are many cells in the aged human, even in a person of a hundred years of age, that are entirely normal and have essentially not aged to a significant degree. The problem in aging is that an increasing percentage of cells in the human body have very serious DNA damage, and it only takes a few of those cells to stimulate what we call bystander effects; they can cause damage to a wide range of cells around that cell, resulting in the manifestations of human aging. But yes, there remain cells, including stem cells, that have pristine DNA, and the selection of those cells to create whole populations of cells is one approach to developing new stem cell therapies.But I believe the most powerful approach is to use those pristine cells as a source of DNA when taken back in this time machine of reprogramming to make all the cell types of the human body pristine, and thereby give the physician the ability to regenerate the inner ear for hearing loss in aging, the retina for macular degeneration, part of the midbrain in Parkinson’s disease, the heart muscle to deal with the number one killer, heart disease, and cartilage, which has no regenerative capacity whatsoever and whose loss is the number one complaint of the elderly, and so on.By taking the cells back in time, both from an aging standpoint and also by taking them back in time in development to make embryonic cells, we hope to unlock early pathways of embryological development that are utilized in primitive vertebrates that allow them to profoundly regenerate tissues, such as re-growing amputated limbs and so on. The clock of cellular aging resides at the very ends of the chromosomes, regions called “telomeres.” Shown here in a blue color are stained human chromosomes, and the telomeric DNA is stained bright yellow.Kent: You may be right about that, but right now the best you can do is one out of six. And as I understand it, the way this technology will be most effectively used is to take somatic cells from individuals and turn them back into embryonic-like cells. What is the chance that you’ll be unable to do so effectively in a particular individual?West: In our paper we manually created these lines, so we only made six cell lines, one of which properly rewound the clock. With the newer robotic platform we are developing, we could increase that number of lines markedly. So, even with no improvements in efficiency, we could increase the number of lines to make a successful outcome for a particular patient highly probable.Fahy: Is the ReCyte™ process patented by BioTime?West: Yes. ReCyte™ is a proprietary technology of BioTime and is based on several issued and pending patents.Fahy: Do you also have exclusive ownership over the process of choosing cells that spontaneously maintain their telomeres and excluding those that don’t?West: We’ve filed for patent protection on the selection process to identify these cell types based on the data in our paper.Fahy: Are there alternative ways of achieving the same effect you’ve been able to obtain? For example, it has been possible to artificially restore telomerase to cells since 1998,3 as you referred to earlier in the interview. Is there any reason iPS cells could not simply be telomerized to correct their defect in telomere maintenance?West: Well, we looked at a telomerized line in our publication, an iPS cell line from Harvard Medical School. That line had, in addition to these germ line genes that make this time machine work embryologically, some added telomerase, in the belief that there may be a problem with the telomere. But those cells still have inappropriately short telomeres. And so what we’ve learned is that telomerase is indeed very important, but merely adding telomerase to the iPS techniques as currently used in most laboratories is not sufficient.Fahy: In the paper, you speculate that maybe the reason the iPS cells are not maintaining their telomeres is that they’re not maintaining their undifferentiated embryonic state over time. Is there any evidence that they might be losing their undifferentiated state?West: All I can say is that what’s so striking about iPS cells is how similar they are to human embryonic stem cells in every respect. If one scans through 30,000 plus genes and looks at the expression of all those genes in human embryonic stem cells and iPS cells they are essentially indistinguishable. It’s very difficult, in fact, to find any differences, and it was hard for us to find what differences might be causing this inefficient resetting of the clock of aging. So the striking thing is their similarity, not that there are problems with the technology. However, we mention in the paper that our survey of widely-studied iPS cell lines may have been influenced by the poor culture of the cells in some laboratories.There have been reports that iPS cells are less likely to make various cell types, like neurons, compared to embryonic stem cells. But it’s important to remember that most of the world is working with iPS cells that are geriatric in terms of aging, and so all of those studies need to be repeated with these new cell lines that have their clock of aging reset properly before any conclusions can be drawn about the normality or abnormality of iPS cells.Fahy: What about the possibility that the cells that are naturally able to restore and maintain their telomeres are those that have DNA damage?West: We looked carefully throughout the genome of these cells using what is called SNP analysis and couldn’t find any evidence of DNA damage such as rearrangements, so for this and other reasons, we don’t have any reason to think the iPS cells that maintain their telomeres do so because of DNA damage the way cancer cells do.A long-standing theory of aging is that our body accumulates cells that have broken chromosomes, serious DNA damage, and damage unrelated to telomere shortening per se. And that almost certainly happens. Nevertheless, we also know that the human body has many trillions of cells that are completely intact, and with modern sequencing technologies it’s now possible for just a few thousand dollars to completely sequence a cell line’s genome. And so I believe it will be commercially feasible to perform a previously unthinkable amount of quality control to be sure that the young cell types that we make by new technologies like ReCyte™ are pristine, young cells as good as the cells we were born from, and not carrying a significant load of genetic damage.Kent: Robert Bradbury and others have talked about plucking pristine stem cells out of a pool of adult stem cells and having the best possible adult stem cells rather than some of them being contaminated or damaged.West: There are many cells in the aged human, even in a person of a hundred years of age, that are entirely normal and have essentially not aged to a significant degree. The problem in aging is that an increasing percentage of cells in the human body have very serious DNA damage, and it only takes a few of those cells to stimulate what we call bystander effects; they can cause damage to a wide range of cells around that cell, resulting in the manifestations of human aging. But yes, there remain cells, including stem cells, that have pristine DNA, and the selection of those cells to create whole populations of cells is one approach to developing new stem cell therapies.But I believe the most powerful approach is to use those pristine cells as a source of DNA when taken back in this time machine of reprogramming to make all the cell types of the human body pristine, and thereby give the physician the ability to regenerate the inner ear for hearing loss in aging, the retina for macular degeneration, part of the midbrain in Parkinson’s disease, the heart muscle to deal with the number one killer, heart disease, and cartilage, which has no regenerative capacity whatsoever and whose loss is the number one complaint of the elderly, and so on.By taking the cells back in time, both from an aging standpoint and also by taking them back in time in development to make embryonic cells, we hope to unlock early pathways of embryological development that are utilized in primitive vertebrates that allow them to profoundly regenerate tissues, such as re-growing amputated limbs and so on.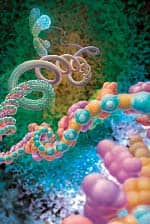 Illustration of a telomere: Telomeres consist of a DNA sequence (TTAGGG) that is repeated over and over again, forming the tips or “caps” of our chromosomes. Breakage or erosion of telomeres as a result of age can trigger cascades of events that lead to tissue degeneration. The telomere is shown being unwound from a chromosome (top left).Fahy: Is there any independent confirmation of your discovery that there is a telomere problem with most iPS cells? Have others found at least any indirect signs that are in agreement with your observations?West: Back in 1999, my old company, Geron, in collaboration with the Roslyn Institute, announced that cloned animals were born old. The thought then was that it would be unthinkable that cloning would be so miraculous that it would reverse both development and aging as well. But in 2000 we published evidence11 that cloning actually does reverse both development and aging, and since that time it’s become established that cloning really does both. So much so that, when iPS researchers saw that telomerase is reactivated in iPS cells, they assumed that this was sufficient for telomeres to be restored, without looking at the telomeres themselves.We were a bit concerned that we were the only ones who had identified that most iPS cells do not rewind the clock of aging, but just recently a group at Advanced Cell Technology published a paper showing that all iPS cell lines they looked at showed accelerated aging when turned into blood-forming cells,16 even though they did not look at telomeres specifically. And in our new paper,14 we cite similar additional indirect evidence from other labs in support of our findings.More recently, a group at Harvard showed an increase in telomere length after reprogramming by iPS technology in cells affected by a premature aging disease caused by a mutation involving one of thetelomerase components.15 And so the scientific community is presently somewhat confused over this issue, but we believe that the answer is that iPS technology is indeed capable of reversing the aging of human cells, it’s just that it’s not 100% of the time, and so it’s necessary to identify the cells that have had their clock of aging rewound.Fahy: How serious of a problem would it be if you used iPS cells that had not had their telomeres restored?West: It all depends on the application. In the case of the heart muscle, if you’re in heart failure and you need new heart muscle regenerated identical to your own so it will be not rejected, I think you could get away with cells that were not embryonic in regard to cellular aging. But for many of the cells of the human body there really is, I believe, a great advantage to giving us back the cells we were born with many decades earlier.An example is blood-forming cells. In aging we can watch telomere length shorten in the blood cells every decade, and the tie-in between that shortening of telomeres and the onset of chronic infectious disease and other immune disorders, like anemias, is pretty well documented. To give us back the blood-forming cells that we were born with, so our immune system would have a robust reservoir of cells capable of fighting infections, would be highly desirable.Fahy: There has been some talk recently about the possibility of transdifferentiation as an alternative to iPS cell technology, in which instead of going back to an embryonic state, you simply convert cell A into target cell B without having to go back in time and then redifferentiate the cell back to target cell B. You just go straight to your target. Do you see that as a significant competitor to iPS cell technology, and do you think that the same issues that you’ve identified for iPS cells will pertain to transdifferentiated cells?West: I believe these master regulatory genes we call transcription factors are able to take cells back to the germ line, but will also be able to take a cell, like a skin cell, directly to a neuron. The advantage of taking cells back to the germ line is that we reset the clock of aging in cells, as well as make all these primitive embryonic lineages that we anticipate will be more effective in regenerating tissue function compared to adult cells.Kent: How do you see regenerative medicine dealing with non-dividing cells such as neurons and muscle cells, which are critically important?West: Non-proliferating cells can become injured or lost as a result of aging, so to extend human longevity, medicine needs to have a way to replace those cells and tissues as well. For example, in Parkinson’s disease, cells are lost in the mid-brain, resulting in the progressive symptoms of the disease, which we can only partially treat today, and the loss of heart muscle cells can progress to heart failure, now a leading cause of death in the United States. The hope of regenerative medicine would be for the first time to add to the doctor’s toolbox products that could allow the regeneration of these cells and tissues to replace those worn out with age and thereby essentially cure these important diseases for the first time.Kent: You just referred to the loss of non-dividing cells as a result of aging, yet you tend to equate aging with telomere shortening, which should not happen in non-dividing cells. Can you clarify what you mean by aging in non-dividing cells?West: I am glad you brought that up, Saul, because there is this debate in the aging research community about the mechanisms of aging, and the critics of the telomere hypothesis use this as one of their arguments. I would point out two things. First, most tissues with non-dividing cells, like the brain and heart, are also full of cells that do divide over time. In the case of the brain, the dividing cells are called astrocytes, and in the heart they are stromal fibroblasts. And so, in my mind it is specious to point to these tissues as evidence against the role of cellular aging in the aging brain and heart since the dividing cells could be important pacemakers. Second, the lack of telomerase activity in non-dividing cells could also impart an inability to repair telomeres broken as a result of normal wear-and-tear. So, we might have degenerative changes occurring in non-dividing cells as a result of the lack of telomerase but not from replicative senescence.Fahy: There are natural precedents for replacement of non-dividing cells in the brain. The brain makes new cells all the time in a process called neurogenesis. This is particularly active in the hippocampus, which is where memories are distributed, so replacing non-dividing cells even in the brain seems to be feasible.Kent: While it’s true that new neurons can be developed in the hippocampus from neural stem cells, which aid in learning and memory, this natural process doesn’t work nearly well enough to prevent the loss of memory and other cognitive functions with aging, or to prevent the development of age-related diseases such as Parkinson’s disease and Alzheimer’s disease. Perhaps the introduction of additional neural stem cells will be able to solve these problems, but isn’t there a risk that if you replace neurons in the brain that contain memories and information regarding the individual’s personality with young, healthy neurons, that these neurons won’t have that information and the identity of the person could be partially or entirely lost?West: Absolutely. The goal of regenerative medicine is, obviously, to both repair the human body and to maintain the identity of the individual in the process, so ultimately we will need to look carefully at this unique problem you’re mentioning regarding specific sites in the brain.Kent: Perhaps rather than replacing dysfunctional neurons outright, we’ll be able to repair them by replacing component parts of these neurons, which might not affect memory encoded in these neurons.West: I think there’s a lot of potential for exactly what you’re suggesting.One of the hallmarks of our time is shifting paradigms in the way we think about aging. Not that many years ago, neurobiologists were set in stone in the belief that the neurons in the human brain do not proliferate, and now we have a complete revolution in thought with scientific evidence that these cells can turn over. This is consistent with what we’re talking about today, regenerative medicine, which could enable doctors to regenerate new, young cells of any kind to replace those worn out with aging.Kent: What evidence do you have to support the idea that once you introduce these young cells into a person’s body, they will rejuvenate the area in which you have injected them or the system which you have tried to rejuvenate?West: In the history of embryology research, there have been scientists who have been transplanting embryonic cells and tissues, oftentimes between different bird species like chick and quail. And what we see there is that these primitive embryonic cells are unusually plastic, by which I mean that they can be scooped up with a syringe or a scalpel and transplanted into corresponding tissue and recognize where they are, and promote scarless regeneration. While the aging human body is progressively dysfunctional and increasingly ravaged with disease processes, the cells that we envision putting into the body are cells with an amazing amount of plasticity that can recognize where they are and what they need to do.A simple way of seeing this is that early in evolution, before telomerase was repressed and a lot of these changes occurred to cause aging, primitive vertebrates maintained their embryonic plasticity, and we can still see this in some animals today. As I alluded to earlier, there are species of salamanders in which you can cut off a leg, you can remove a large chunk of their heart or even of their brain, and the tissues will regenerate. An arm will grow back right where it was cut off, and you’ll have a new functioning limb with blood vessels and nerves and muscles and bones and cartilage, all in their proper places. That is the type of plasticity and regenerative capacity that these embryonic cells have, and it is the reason Bill Haseltine christened this field regenerative medicine.Kent: Another possibility is that there could be a risk of getting cancer as a result of using immortal iPS cells. Illustration of a telomere: Telomeres consist of a DNA sequence (TTAGGG) that is repeated over and over again, forming the tips or “caps” of our chromosomes. Breakage or erosion of telomeres as a result of age can trigger cascades of events that lead to tissue degeneration. The telomere is shown being unwound from a chromosome (top left).Fahy: Is there any independent confirmation of your discovery that there is a telomere problem with most iPS cells? Have others found at least any indirect signs that are in agreement with your observations?West: Back in 1999, my old company, Geron, in collaboration with the Roslyn Institute, announced that cloned animals were born old. The thought then was that it would be unthinkable that cloning would be so miraculous that it would reverse both development and aging as well. But in 2000 we published evidence11 that cloning actually does reverse both development and aging, and since that time it’s become established that cloning really does both. So much so that, when iPS researchers saw that telomerase is reactivated in iPS cells, they assumed that this was sufficient for telomeres to be restored, without looking at the telomeres themselves.We were a bit concerned that we were the only ones who had identified that most iPS cells do not rewind the clock of aging, but just recently a group at Advanced Cell Technology published a paper showing that all iPS cell lines they looked at showed accelerated aging when turned into blood-forming cells,16 even though they did not look at telomeres specifically. And in our new paper,14 we cite similar additional indirect evidence from other labs in support of our findings.More recently, a group at Harvard showed an increase in telomere length after reprogramming by iPS technology in cells affected by a premature aging disease caused by a mutation involving one of thetelomerase components.15 And so the scientific community is presently somewhat confused over this issue, but we believe that the answer is that iPS technology is indeed capable of reversing the aging of human cells, it’s just that it’s not 100% of the time, and so it’s necessary to identify the cells that have had their clock of aging rewound.Fahy: How serious of a problem would it be if you used iPS cells that had not had their telomeres restored?West: It all depends on the application. In the case of the heart muscle, if you’re in heart failure and you need new heart muscle regenerated identical to your own so it will be not rejected, I think you could get away with cells that were not embryonic in regard to cellular aging. But for many of the cells of the human body there really is, I believe, a great advantage to giving us back the cells we were born with many decades earlier.An example is blood-forming cells. In aging we can watch telomere length shorten in the blood cells every decade, and the tie-in between that shortening of telomeres and the onset of chronic infectious disease and other immune disorders, like anemias, is pretty well documented. To give us back the blood-forming cells that we were born with, so our immune system would have a robust reservoir of cells capable of fighting infections, would be highly desirable.Fahy: There has been some talk recently about the possibility of transdifferentiation as an alternative to iPS cell technology, in which instead of going back to an embryonic state, you simply convert cell A into target cell B without having to go back in time and then redifferentiate the cell back to target cell B. You just go straight to your target. Do you see that as a significant competitor to iPS cell technology, and do you think that the same issues that you’ve identified for iPS cells will pertain to transdifferentiated cells?West: I believe these master regulatory genes we call transcription factors are able to take cells back to the germ line, but will also be able to take a cell, like a skin cell, directly to a neuron. The advantage of taking cells back to the germ line is that we reset the clock of aging in cells, as well as make all these primitive embryonic lineages that we anticipate will be more effective in regenerating tissue function compared to adult cells.Kent: How do you see regenerative medicine dealing with non-dividing cells such as neurons and muscle cells, which are critically important?West: Non-proliferating cells can become injured or lost as a result of aging, so to extend human longevity, medicine needs to have a way to replace those cells and tissues as well. For example, in Parkinson’s disease, cells are lost in the mid-brain, resulting in the progressive symptoms of the disease, which we can only partially treat today, and the loss of heart muscle cells can progress to heart failure, now a leading cause of death in the United States. The hope of regenerative medicine would be for the first time to add to the doctor’s toolbox products that could allow the regeneration of these cells and tissues to replace those worn out with age and thereby essentially cure these important diseases for the first time.Kent: You just referred to the loss of non-dividing cells as a result of aging, yet you tend to equate aging with telomere shortening, which should not happen in non-dividing cells. Can you clarify what you mean by aging in non-dividing cells?West: I am glad you brought that up, Saul, because there is this debate in the aging research community about the mechanisms of aging, and the critics of the telomere hypothesis use this as one of their arguments. I would point out two things. First, most tissues with non-dividing cells, like the brain and heart, are also full of cells that do divide over time. In the case of the brain, the dividing cells are called astrocytes, and in the heart they are stromal fibroblasts. And so, in my mind it is specious to point to these tissues as evidence against the role of cellular aging in the aging brain and heart since the dividing cells could be important pacemakers. Second, the lack of telomerase activity in non-dividing cells could also impart an inability to repair telomeres broken as a result of normal wear-and-tear. So, we might have degenerative changes occurring in non-dividing cells as a result of the lack of telomerase but not from replicative senescence.Fahy: There are natural precedents for replacement of non-dividing cells in the brain. The brain makes new cells all the time in a process called neurogenesis. This is particularly active in the hippocampus, which is where memories are distributed, so replacing non-dividing cells even in the brain seems to be feasible.Kent: While it’s true that new neurons can be developed in the hippocampus from neural stem cells, which aid in learning and memory, this natural process doesn’t work nearly well enough to prevent the loss of memory and other cognitive functions with aging, or to prevent the development of age-related diseases such as Parkinson’s disease and Alzheimer’s disease. Perhaps the introduction of additional neural stem cells will be able to solve these problems, but isn’t there a risk that if you replace neurons in the brain that contain memories and information regarding the individual’s personality with young, healthy neurons, that these neurons won’t have that information and the identity of the person could be partially or entirely lost?West: Absolutely. The goal of regenerative medicine is, obviously, to both repair the human body and to maintain the identity of the individual in the process, so ultimately we will need to look carefully at this unique problem you’re mentioning regarding specific sites in the brain.Kent: Perhaps rather than replacing dysfunctional neurons outright, we’ll be able to repair them by replacing component parts of these neurons, which might not affect memory encoded in these neurons.West: I think there’s a lot of potential for exactly what you’re suggesting.One of the hallmarks of our time is shifting paradigms in the way we think about aging. Not that many years ago, neurobiologists were set in stone in the belief that the neurons in the human brain do not proliferate, and now we have a complete revolution in thought with scientific evidence that these cells can turn over. This is consistent with what we’re talking about today, regenerative medicine, which could enable doctors to regenerate new, young cells of any kind to replace those worn out with aging.Kent: What evidence do you have to support the idea that once you introduce these young cells into a person’s body, they will rejuvenate the area in which you have injected them or the system which you have tried to rejuvenate?West: In the history of embryology research, there have been scientists who have been transplanting embryonic cells and tissues, oftentimes between different bird species like chick and quail. And what we see there is that these primitive embryonic cells are unusually plastic, by which I mean that they can be scooped up with a syringe or a scalpel and transplanted into corresponding tissue and recognize where they are, and promote scarless regeneration. While the aging human body is progressively dysfunctional and increasingly ravaged with disease processes, the cells that we envision putting into the body are cells with an amazing amount of plasticity that can recognize where they are and what they need to do.A simple way of seeing this is that early in evolution, before telomerase was repressed and a lot of these changes occurred to cause aging, primitive vertebrates maintained their embryonic plasticity, and we can still see this in some animals today. As I alluded to earlier, there are species of salamanders in which you can cut off a leg, you can remove a large chunk of their heart or even of their brain, and the tissues will regenerate. An arm will grow back right where it was cut off, and you’ll have a new functioning limb with blood vessels and nerves and muscles and bones and cartilage, all in their proper places. That is the type of plasticity and regenerative capacity that these embryonic cells have, and it is the reason Bill Haseltine christened this field regenerative medicine.Kent: Another possibility is that there could be a risk of getting cancer as a result of using immortal iPS cells. Michael West, PhD—CEO, BioTime, Inc.West: That’s possible. Some scientists hypothesize that in order to live as long as humans live without dying of cancer, evolution selected to turn off the immortalizing gene telomerase in virtually all cells of the human body. The result is that it gave us a genetic limit to our life span, but it also limited our exposure to cancer. Once we’ve reproduced and raised our babies, we’ve helped to continue the species, and it is okay from an evolutionary standpoint to die from old age.But it’s important to remember that the iPS cells only are immortal in the germ line state. Once we turn them into the early cell lineages of the human body, telomerase is shut off.Kent: Severe calorie restriction extends maximum life span, which indicates that aging has been slowed, but postpones the incidence of cancer as well. So both problems can be addressed simultaneously.West: I share your optimism. I believe that through new technologies aimed at targeting and destroying tumors combined with a more complete understanding of the connections between telomere shortening and cancer, we can find ways to walk that tightrope. If we eliminate the major causes of aging and dramatically improve the quality of human aging or even human life span, it’s almost a certainty that we will increasingly then have to face the problems of cancer. That’s just the reality of human biology.Kent: If we had a chance of slowing or reversing aging and increasing our maximum healthy life span in exchange for an added risk of cancer, I think many people would likely take that risk because cancer in many cases is both preventable and curable, but so far aging is not.Fahy: Since all of the adult-onset forms of cancer are probably caused by telomere shortening rather than from having telomeres that are too long,17 it seems that nature’s use of telomere shortening as an anti-cancer device may have been overdone! Also, immortal stem cells already exist in some places in the body, and yet most people don’t die of cancer.Kent: When do you anticipate that the initial therapies based on iPS cell technology will be available to humans?West: In the United States, with its stringent regulatory burden of approval, it could take many years. In other countries, such as China and India, you have very sophisticated and large medical establishments and even larger aging populations than the US. In the case of China, the post-Mao baby-boom generation is significantly larger than the post World War II baby-boom generation in the US. I believe that these technologies will be implemented in a fraction of the time there than in the United States. But it’s important, again, to emphasize that nowhere in the world are these technologies available today. I would urge people not to rush off and seek therapies that have not been proven safe and effective. Michael West, PhD—CEO, BioTime, Inc.West: That’s possible. Some scientists hypothesize that in order to live as long as humans live without dying of cancer, evolution selected to turn off the immortalizing gene telomerase in virtually all cells of the human body. The result is that it gave us a genetic limit to our life span, but it also limited our exposure to cancer. Once we’ve reproduced and raised our babies, we’ve helped to continue the species, and it is okay from an evolutionary standpoint to die from old age.But it’s important to remember that the iPS cells only are immortal in the germ line state. Once we turn them into the early cell lineages of the human body, telomerase is shut off.Kent: Severe calorie restriction extends maximum life span, which indicates that aging has been slowed, but postpones the incidence of cancer as well. So both problems can be addressed simultaneously.West: I share your optimism. I believe that through new technologies aimed at targeting and destroying tumors combined with a more complete understanding of the connections between telomere shortening and cancer, we can find ways to walk that tightrope. If we eliminate the major causes of aging and dramatically improve the quality of human aging or even human life span, it’s almost a certainty that we will increasingly then have to face the problems of cancer. That’s just the reality of human biology.Kent: If we had a chance of slowing or reversing aging and increasing our maximum healthy life span in exchange for an added risk of cancer, I think many people would likely take that risk because cancer in many cases is both preventable and curable, but so far aging is not.Fahy: Since all of the adult-onset forms of cancer are probably caused by telomere shortening rather than from having telomeres that are too long,17 it seems that nature’s use of telomere shortening as an anti-cancer device may have been overdone! Also, immortal stem cells already exist in some places in the body, and yet most people don’t die of cancer.Kent: When do you anticipate that the initial therapies based on iPS cell technology will be available to humans?West: In the United States, with its stringent regulatory burden of approval, it could take many years. In other countries, such as China and India, you have very sophisticated and large medical establishments and even larger aging populations than the US. In the case of China, the post-Mao baby-boom generation is significantly larger than the post World War II baby-boom generation in the US. I believe that these technologies will be implemented in a fraction of the time there than in the United States. But it’s important, again, to emphasize that nowhere in the world are these technologies available today. I would urge people not to rush off and seek therapies that have not been proven safe and effective.  Gregory M. Fahy, PhD Gregory M. Fahy, PhDChief Scientific Officer and Vice President, 21st Century MedicineSaul Kent Co-Founder, Life Extension FoundationA tsunami of aging is coming our way, which is going to be disastrous in its consequences, both to the individuals in need of healthcare and to the economies of countries that will be faced with escalating healthcare costs. In my opinion, we now have the science and technologies to prepare for this tsunami by developing new medical therapies within the next 10 years to reverse age-related degenerative diseases and profoundly influence the course of aging itself.In the face of this impending healthcare and economic crisis, many gerontologists are reaching out for capital to fund their research. To advance these novel therapies through regulatory approval will require significant capital expenditure. The day that these technologies can be used to treat human beings is entirely dependent on the number of scientists that can be employed to move development forward.I want to take this opportunity to thank the Life Extension Foundation for their financial help in the early stages of this project. Now it’s time for Congress to fund basic research in aging. The looming baby-boom population threatens to break the bank. Our future is in the balance.Fahy: Thank you, Dr. West, for this extremely important and strategic look into the future of therapies to combat aging. |
| References |
| 1. Cooke HJ, Smith BA. Variability at the telomeres of the human X/Y pseudoautosomal region. Cold Spring Harb Symp Quant Biol. 1986;51 Pt 1:213-9.2. Olovnikov AM. Principle of marginotomy in template synthesis of polynucleotides. Dokl Akad Nauk SSSR. 1971;201(6):1496-9.3. Bodnar AG, Ouellette M, Frolkis M, et al. Extension of cell lifespan by introduction of telomerase into normal human cells. Science. 1998 Jan 16;279(5349):349-52.4. Thomson JA, Itskovitx-Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998 Nov 6;282(5391):1145-7.5. Shamblott MJ, Axelman J, Wang S, et al. Derivation of pluripotent stem cells from cultured human primordial germ cells. Proc Natl Acad Sci. 1998 Nov 10;95(23):13726-31.6. Cibelli JB, Kiessling AA, Cunniff K, Richards C, Lanza RP, West MD. Somatic cell nuclear transfer in humans: Pronuclear and early embryonic development. J Regen Med. 2001;2:25-31.7. Lanza RP, Cibelli JB, West MD. Human therapeutic cloning. Nat Med.1999;Sep5(9):975-7.8. Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007 Nov 30;131(5):861-72.9. Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007 Dec 21;318(5858):1917-20.10. Shiels PG, Kind AJ, Campbell KH, et al. Analysis of telomere lengths in cloned sheep. Nature. 1999 Sep;399(6734):316-7.11. Lanza RP, Cibelli JB, Blackwell C, et al. Extension of cell life-span and telomere length in animals cloned from senescent somatic cells. Science. 2000 Apr 28;288(5466):665-9.12. Kishigami S, Wakayama S, Hosoi Y, et al. Somatic cell nuclear transfer: Infinite reproduction of a unique diploid genome. Exp Cell Res. 2008 Jun 10;314(9):1945-50.13. Jang G, Hong SG, Oh HJ, et al. A cloned toy poodle produced from somatic cells derived from an aged female dog. Theriogenology. 2008 Mar 15;69(5):556-63.14. Vaziri, H. Chapman, KB, Guigova, A, et al. Spontaneous reversal of the developmental aging of normal human cells following transcriptional reprogramming. Regen Med. 2010 Mar 16.15. Agarwal S, Loh YH, McLoughlin EM, et al. Telomere elongation in induced pluripotent stem cells from dyskeratosis congenital patients. Nature. 2010 Mar 11;464(7286):292-6.16. Feng Q, Lu SJ, Klimanskaya I, et al. Hemangioblastic derivatives from human induced pluripotent stem cells exhibit limited expansion an early senescence. Stem Cells Online. 2010 Feb 12.17. DePinho RA. The age of cancer. Nature. 2000 Nov 9;408(6809):248-54. |




